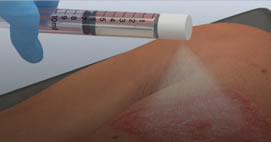
ReCell, a regenerative skin-cell solution, is the first treatment of its kind approved by the US Food and Drug Administration for growing back skin.
“You just spray it on the patient, on the burned area,” said Chris Houchens of the federal government’s Biomedical Advanced Research and Development Authority, or BARDA, which helped pay for its development.
In the United States alone, roughly half a million people have to seek medical treatment for their burns each year. The most traditional form of treatment came in the form of skin grafts. However, those require more pain on the part of the patient, as Doctors have to cut away and remove healthy skin elsewhere on the body to cover the burns.
ReCell reduces how much skin has to be removed over the burned surface before treatment, the company explained. It uses enzymes to break down those layers of skin from a piece of tissue. It then mixes those tissue cells into a liquid that’s applied to the skin using a simple, low-tech spray.
ReCell is definitely not the only solution that’s been in the works. It’s simply the first one to gain FDA approval.
In 2009, MIT researchers made headlines when they proposed a similar spray-on skin. AVITA Medical competitor RenovaCare also created the SkinGun.
Close to half a million people get burns so bad that they need medical treatment, according to the American Burn Association and the Centers for Disease Control and Prevention.